
By this means, the Bland-Altman limits of agreement and the applied statistical hypothesis test are by definition interrelated. In the following, we will base the statistical analysis strategy on the true (but unknown) population standard deviation of the paired differences. 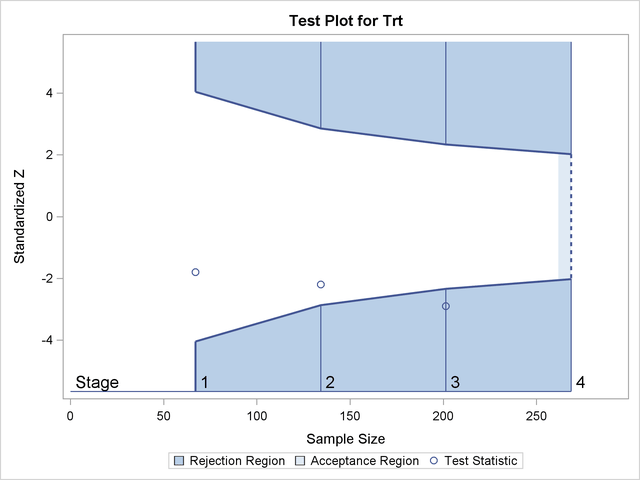
Implicitly, it is assumed that the paired differences of the whole target population from which the sample was taken follow a normal distribution then, the Bland-Altman limits of agreement comprise, on average, 95% of all observations according to the 68-95-99.7 rule and can be interpreted as prediction interval. These limits consist of the mean difference of the paired measurements ± 1.96 times the sample standard deviation of these differences. The agreement of paired, quantitative measurements in method comparison or observer variation studies is often assessed with Bland-Altman plots with respective limits of agreement. The purpose of this paper was to demonstrate a group-sequential analysis strategy in an intra-observer study on quantitative FDG-PET/CT measurements, illuminating the possibility of early trial termination which implicates significant potential time and resource savings. While group-sequential testing is regularly done in pivotal clinical trials with therapeutic intent, it is much less common in diagnostic trials. This built-in possibility requires adjustment of analyses for multiple testing, for which suitable approaches are at hand. In contrast, group-sequential trial designs are hallmarked by predefinition of number, time points, and stopping rules of interim analyses to enable the trial to be terminated early due to either fertility or futility should the trial develop against former expectations. Most clinical trials employ fixed-sample designs in which the data of all patients are collected and first examined at the end of the study. Planning, conduct, analysis, and report of clinical trials require comprehensive resources. Group-sequential testing may have a place in accuracy and agreement studies. The testing strategy must, though, be defined at the planning stage, and sample sizes must be reasonably large at interim analysis to ensure robustness against single outliers. Group-sequential testing may enable early stopping of a trial, allowing for potential time and resource savings. Other partitions did not suggest early stopping after adjustment for multiple testing due to one influential outlier and our small sample size. 
When performing interim analyses with one third and two thirds of patients, sufficient agreement could be concluded after the first interim analysis and the final analysis. The fixed-sample analysis ( N = 45) was compared with the group-sequential analysis strategies comprising one (at N = 23), two (at N = 15, 30), or three interim analyses (at N = 11, 23, 34), respectively, which were defined post hoc. Differences in SUVmax were assumed to be normally distributed, and sequential one-sided hypothesis tests on the population standard deviation of the differences against a hypothesised value of 1.5 were performed, employing an alpha spending function. Primary lesion maximum standardised uptake value (SUVmax) was determined twice from preoperative FDG-PET/CTs in 45 ovarian cancer patients. 
Group-sequential testing is widely used in pivotal therapeutic, but rarely in diagnostic research, although it may save studies, time, and costs.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
